Quantum Numbers for Atoms - Chemistry LibreTexts
5 (732) · $ 15.00 · In stock
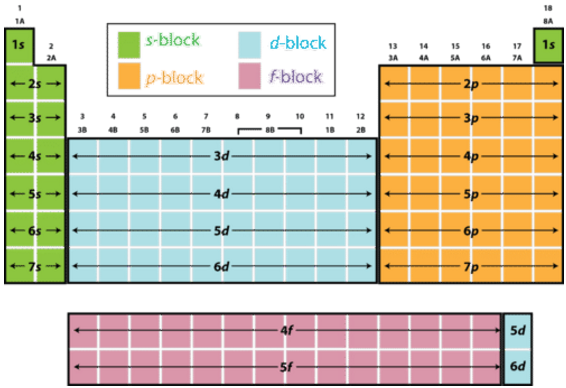
A total of four quantum numbers are used to describe completely the movement and trajectories of each electron within an atom. The combination of all quantum numbers of all electrons in an atom is …
A total of four quantum numbers are used to describe completely the movement and trajectories of each electron within an atom. The combination of all quantum numbers of all electrons in an atom is described by a wave function that complies with the Schrödinger equation. Each electron in an atom has a unique set of quantum numbers; according to the Pauli Exclusion Principle, no two electrons can share the same combination of four quantum numbers.

2.7: Electron Spin- A Fourth Quantum Number - Chemistry LibreTexts

9.A Condensed Matter Physics (Answers) - Physics LibreTexts

Las shs gen.chem-melc_1_q2_week-1
Question #51981

Science Activity Sheet: Quarter 2 - MELC 1 Week 1, PDF, Atomic Orbital
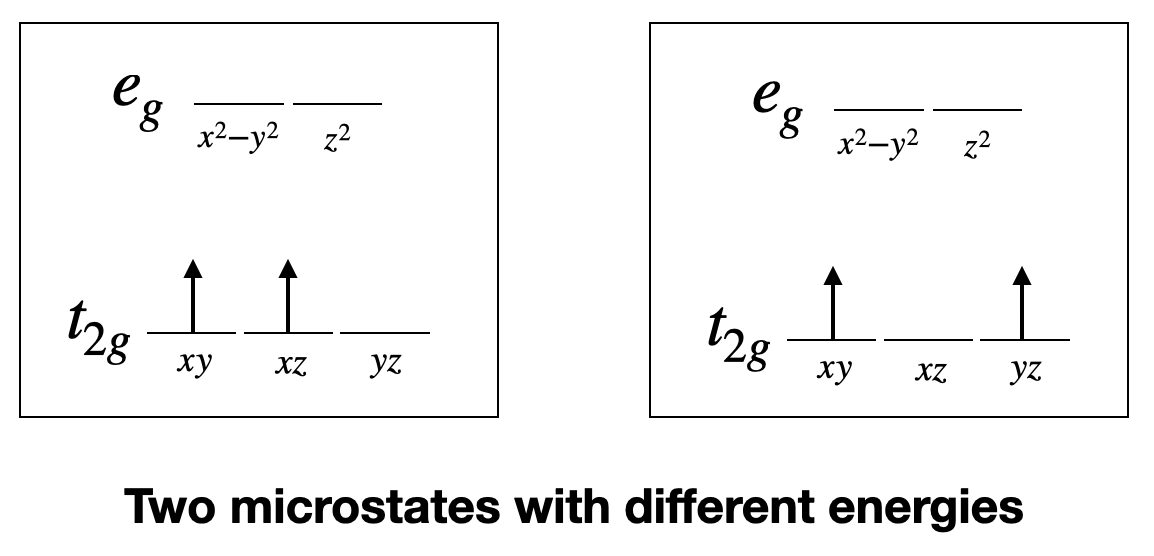
13.1: Quantum Numbers of Multielectron Atoms - Chemistry LibreTexts

The Quantum Numbers Understanding Atomic Structure

Impressions: Robinson's Brutus Awards For 2015, Part, 42% OFF

Family Of V(III)-Tristhiolato Complexes Relevant To, 46% OFF

Subatomic particle, Definition, Examples, & Classes

Impressions: Robinson's Brutus Awards For 2015, Part, 42% OFF

Atom Definition, Structure, History, Examples, Diagram, & Facts
This Little-Known Quantum Rule Makes Our Existence Possible

Impressions: Robinson's Brutus Awards For 2015, Part, 42% OFF